Comparing Data Sets
Help Questions
ACT Science › Comparing Data Sets
Based on Figures 1 and 2, which substance behaves most similarly to Oxygen gas (O₂) in terms of how its solubility changes with temperature?
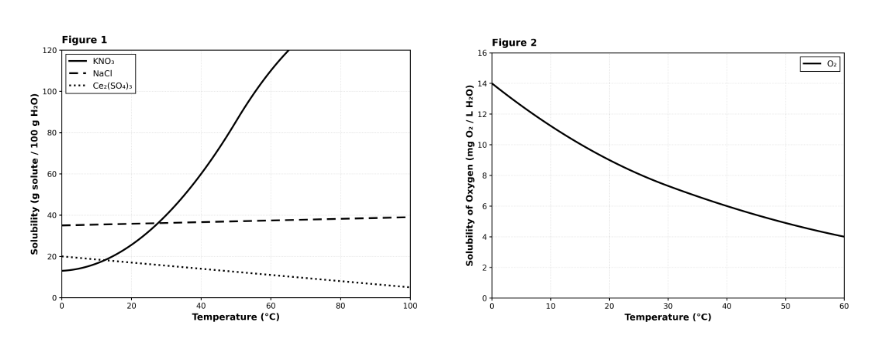
KNO₃
NaCl
Ce₂(SO₄)₃
None of the solids behave like the gas.
Explanation
This is a cross-figure synthesis question requiring you to compare trends across two different graphs. Figure 2 shows O₂ solubility decreases as temperature increases (downward-sloping curve). Looking at Figure 1, you need to find which solid also shows decreasing solubility with increasing temperature. KNO₃ increases dramatically, NaCl stays nearly flat, but Ce₂(SO₄)₃ decreases from 20 g at 0°C to 5 g at 100°C. This downward trend matches O₂'s behavior. Choice C is correct. Choices A and B show opposite or neutral trends. Choice D is incorrect because Ce₂(SO₄)₃ does behave similarly. Pro tip: For similarity questions, identify the key pattern (here: decreasing with temperature) and find the match.
Based on Table 2, which surface material created the greatest resistance (friction) to the movement of the block?
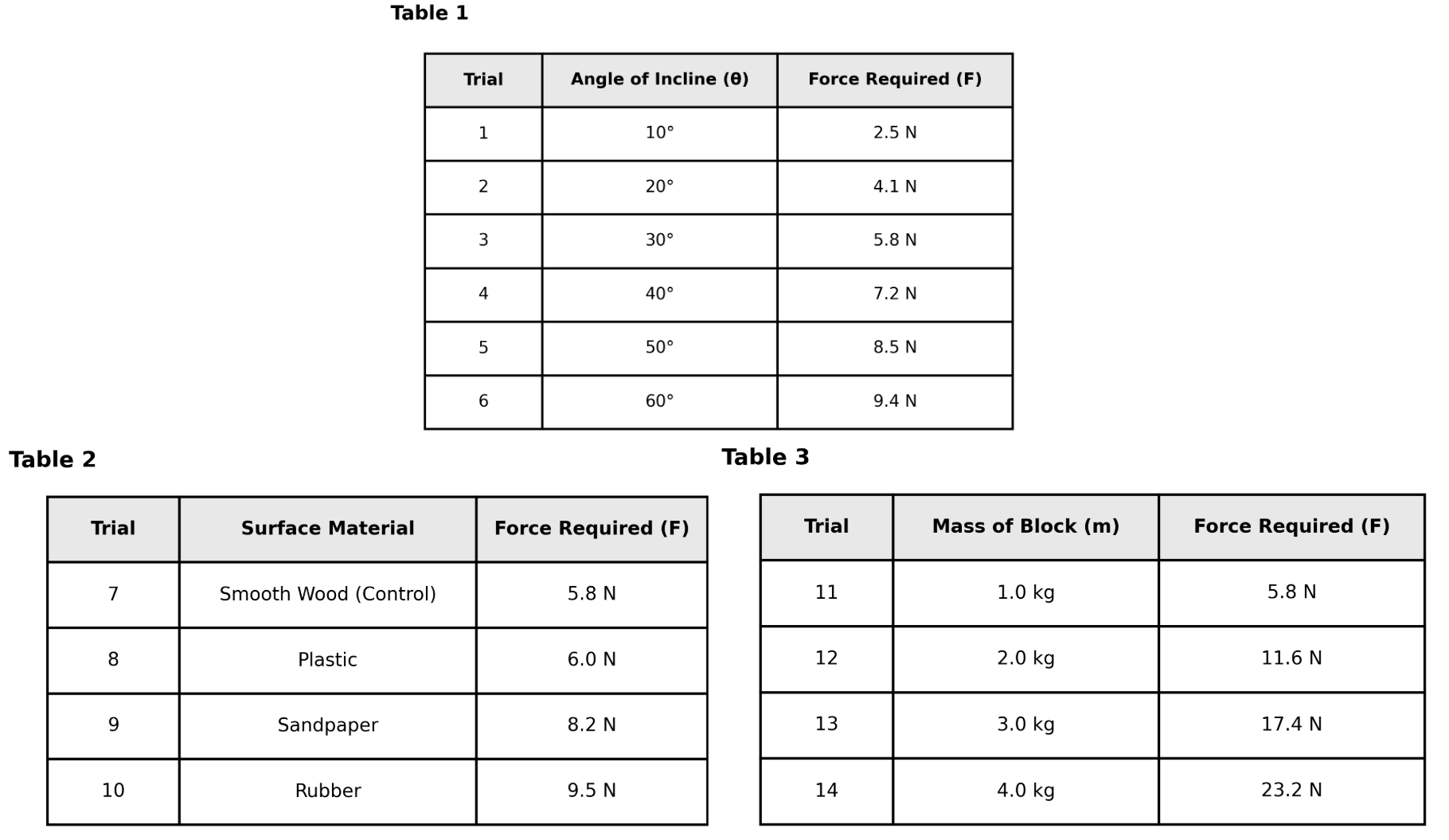
Plastic
Sandpaper
Rubber
Smooth Wood
Explanation
This is a data comparison question requiring you to understand that higher force indicates higher friction. Table 2 shows forces for different surfaces: Smooth Wood (5.8 N), Plastic (6.0 N), Sandpaper (8.2 N), and Rubber (9.5 N). Since friction opposes motion and requires more force to overcome, the surface with the highest force has the greatest friction. Rubber at 9.5 N is highest. Choice D is correct. The other choices show lower forces and therefore lower friction. Pro tip: In physics contexts, resistance to motion correlates with higher force needed to maintain constant velocity.
A pharmacology lab monitored a drug in blood plasma. Table A lists measured concentration (mg/L) at several times after dosing. Table B lists percent of the initial concentration at the same times, calculated as $100\times C_t/C_0$. Compare what Table B changes relative to Table A.
Table B reports new measured mg/L values from a different dose, while Table A reports percentages.
Table A reports percent remaining, while Table B reports time in hours converted to minutes.
Table B expresses each time point as percent of the initial concentration, normalizing Table A’s mg/L values.
Both tables report absolute concentration in mg/L, but Table B uses a different chromatography method.
Explanation
Table B normalizes Table A's absolute concentration measurements to percentages relative to the initial value at the same time points. Table A reports drug concentration in mg/L measured by chromatography at four times after dosing; Table B reports percentage of initial concentration (% of C₀) calculated as 100 × (C_t / C₀). This normalization matters because it focuses on relative decline, enabling comparisons across varying initial doses or subjects. A distractor might reverse which table has absolute versus relative values, but the captions specify A as measured mg/L and B as derived percentages.
A physics lab investigated motion down an incline. Table A shows measured position versus time from video tracking. Table B shows calculated velocity versus time computed from the position data using $
v=\Delta x/\Delta t$. Determine the best description of the relationship.
Table B lists position values measured by a different camera, while Table A lists calculated velocities.
Table A reports velocity in m/s, while Table B reports the same velocity in cm/s.
Table A and Table B both report acceleration, but Table B uses smaller time steps.
Table B lists velocity derived from Table A’s position-time data using differences over equal time intervals.
Explanation
Table B derives its velocity data directly from the position-time measurements in Table A using a calculation over uniform intervals. Table A lists cart position in meters (m) at specific times in seconds (s) from video tracking software; Table B lists velocity in m/s calculated as the change in position divided by the time step for corresponding times. This transformation matters because it shows how raw position data can be processed to reveal motion characteristics like speed, teaching the importance of derived quantities in kinematics. A distractor might swap which table has measured versus calculated data, but the captions confirm A as direct measurements and B as derived.
A city air-quality project monitored particulate matter near two locations.
Table A shows hourly measured PM 2.5 concentrations from a roadside sensor on one weekday.
Table B shows daily averaged PM 2.5 from a rooftop station over the same week.
Compare how the two tables differ in time scale and sampling location.
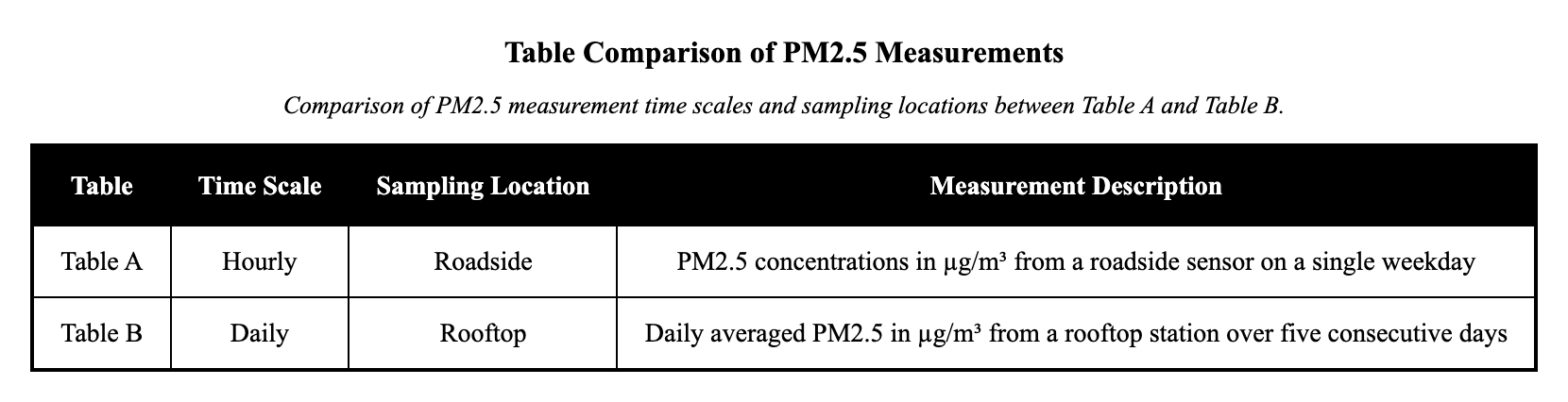
Table A shows daily averages from a rooftop site, while Table B shows hourly roadside spikes.
Table A reports hourly roadside PM$_{2.5}$ for one day, while Table B reports daily rooftop averages across several days.
Table A reports PM${10}$, while Table B reports PM${2.5}$ using the same monitoring method.
Table A and Table B both show hourly PM$_{2.5}$, but in different units.
Explanation
The primary relationship between Table A and Table B is their differing time scales and sampling locations for PM${2.5}$ measurements. Table A shows hourly PM${2.5}$ concentrations in µg/m³ from a roadside sensor on a single weekday, capturing short-term traffic-related spikes; Table B shows daily averaged PM$_{2.5}$ in µg/m³ from a rooftop station over five consecutive days, smoothing out hourly variations to represent overall daily exposure. This difference matters because it affects how the data reflect air quality—short-term peaks versus broader trends—requiring careful attention to context when comparing pollution levels. A distractor might reverse the locations or time scales, but the captions clearly specify roadside hourly for A and rooftop daily for B.
A nutrition study compared two ways of reporting sugar in beverages. Table A lists measured sugar mass per serving in grams (g) from lab analysis. Table B lists calculated sugar concentration in grams per 100 mL, using each drink’s serving volume. Identify the key difference between the tables.
Table A reports sugar per serving measured in grams, while Table B reports calculated sugar per 100 mL normalized by volume.
Table A reports sugar concentration per 100 mL, while Table B reports calories per serving from labels.
Table A reports calculated sugar, while Table B reports measured sugar using a different laboratory method.
Both tables report measured sugar per serving, but Table B uses ounces instead of grams.
Explanation
The key difference is that Table A reports absolute sugar mass per serving, while Table B normalizes it to concentration per volume using calculations. Table A shows measured sugar in grams per serving (g/serving) from lab analysis for four beverages with varying serving sizes; Table B shows calculated sugar in grams per 100 mL by dividing mass by volume and scaling. This normalization matters because it enables fair comparisons across drinks regardless of portion size, focusing on density rather than total intake. A distractor could claim both are measured in similar ways, but Table B's values are explicitly derived from A's data.
An oceanography lab compared salinity measurements from two instruments. Table A shows salinity measured in practical salinity units (PSU) using a conductivity probe. Table B shows chloride concentration measured in g/L using a titration method at the same depths. Identify the primary difference in what is being measured and how.
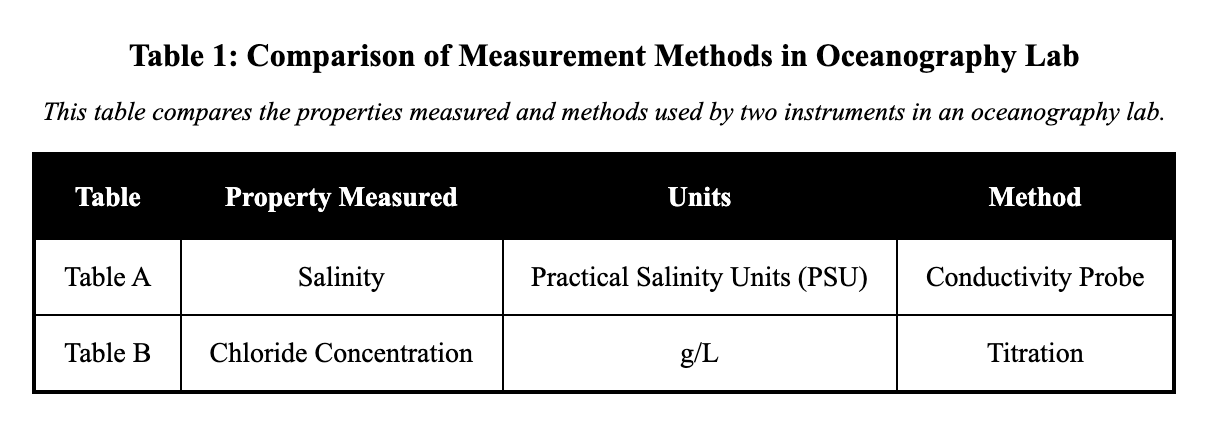
Both tables measure chloride, but Table A reports daily averages and Table B reports hourly spikes.
Table A measures salinity (PSU) with a conductivity probe, while Table B measures chloride (g/L) by titration at the same depths.
Table A measures chloride (g/L) by titration, while Table B measures salinity (PSU) with a conductivity probe.
Both tables measure salinity in PSU, but Table B uses a different depth scale in centimeters.
Explanation
The main difference lies in the specific property measured and the method used, even at the same depths. Table A reports salinity in practical salinity units (PSU) using a conductivity probe during a cast; Table B reports chloride concentration in g/L using lab titration of samples. This distinction matters because salinity estimates total salts via conductivity, while chloride measures one component chemically, affecting how data are interpreted for ocean properties. A distractor could swap the methods or variables, but the captions specify conductivity for salinity in A and titration for chloride in B.
Based on Figures 1 and 2, the sharp increase in density at a depth of 5,100 km corresponds to which change in P-wave velocity?
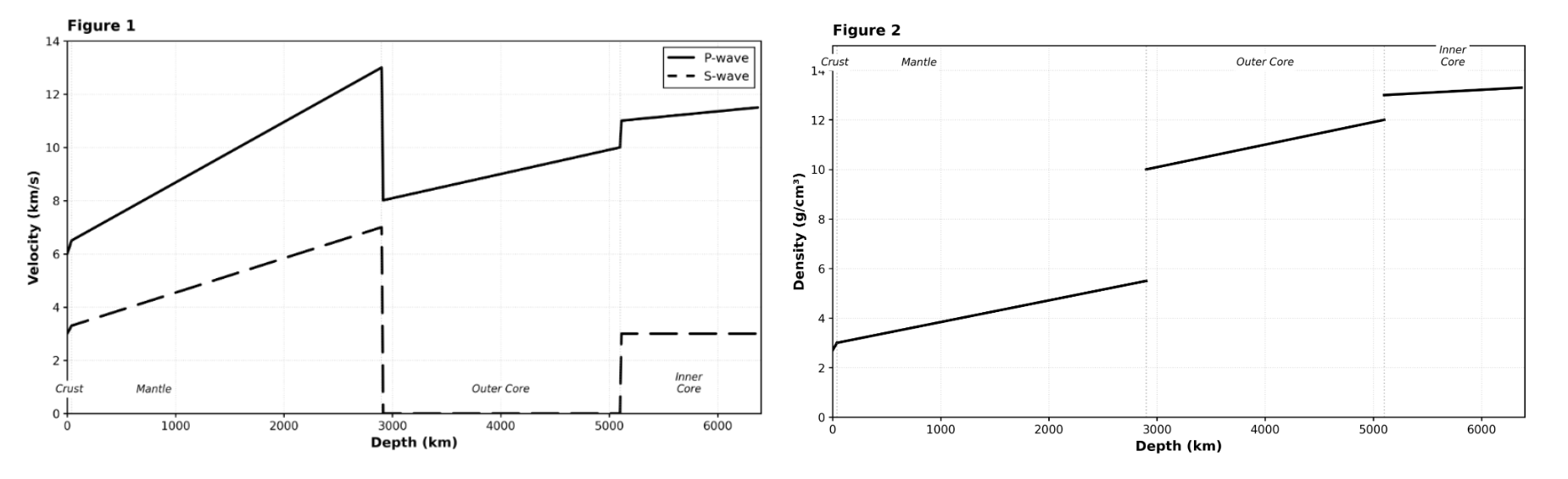
A decrease from 10 km/s to 0 km/s.
An increase from 10 km/s to 11 km/s.
An increase from 8 km/s to 13 km/s.
A decrease from 13 km/s to 8 km/s.
Explanation
This is a cross-figure synthesis question. You need to examine both figures at the same depth (5,100 km, the Outer Core/Inner Core boundary). Figure 2 shows density jumping from 12.0 to 13.0 g/cm³. Figure 1 shows the P-wave curve jumping from approximately 10 km/s (end of Outer Core) to approximately 11 km/s (beginning of Inner Core). Choice C correctly describes this change. Choices A and D describe changes at the wrong boundary (2,900 km). Choice B describes S-wave behavior, not P-wave. Pro tip: For cross-figure questions, carefully identify the same location on both graphs.
Consider the plant in Chamber 3 and the plant in Chamber 7. Which of the following statements about these two plants is most accurate?
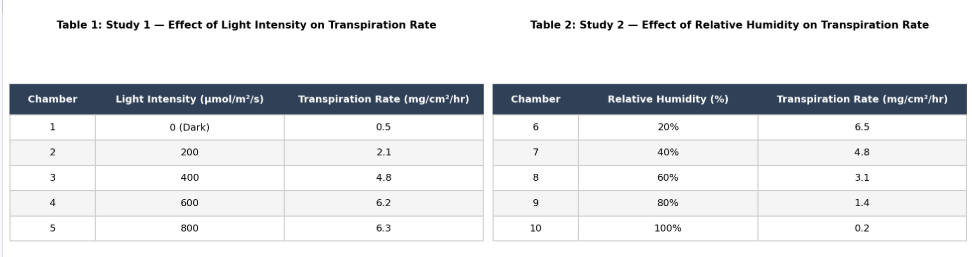
The plant in Chamber 7 was exposed to a higher light intensity than the plant in Chamber 3.
Both plants were exposed to the same environmental conditions (40% humidity and 400 μmol/m²/s light) and had the same transpiration rate of 4.8 mg/cm²/hr.
The plant in Chamber 3 had a transpiration rate of 6.5, while the plant in Chamber 7 had a transpiration rate of 4.8.
The plant in Chamber 3 experienced a higher temperature than the plant in Chamber 7.
Explanation
The correct answer is C. Chamber 3 was in Study 1: light intensity was varied, humidity was constant at 40%, and Chamber 3 received 400 μmol/m²/s light, producing a transpiration rate of 4.8. Chamber 7 was in Study 2: humidity was varied, light intensity was constant at 400 μmol/m²/s, and Chamber 7 had 40% humidity, also producing 4.8. Both plants were therefore exposed to identical conditions (400 light, 40% humidity) and produced identical transpiration rates. A is wrong — temperature was held constant at 22°C in both studies. B is wrong — both chambers received 400 μmol/m²/s. D is wrong — 6.5 corresponds to Chamber 6 (20% humidity), not Chamber 3. Pro tip: Cross-study comparison questions require identifying the controlled variables in each study and checking whether they match the comparison point.
Which of the following best describes the relationship between the two data tables? Table A shows the temperature in Celsius of a chemical reaction over time, while Table B shows the pressure in atmospheres of the same reaction under identical conditions.
Both tables measure the same variable under different conditions.
Table A shows pressure, while Table B shows velocity.
Table A and B use different units for the same measurements.
Table A measures temperature; Table B measures pressure.
Explanation
Table A measures temperature while Table B measures pressure—they track completely different physical variables. Table A shows temperature data in Celsius for a chemical reaction over time, while Table B shows pressure data in atmospheres for the same reaction under identical conditions. This demonstrates that the same experimental system can be monitored for multiple different properties simultaneously. The key distinction is that these are fundamentally different types of measurements, not variations of the same variable.