Organic Concepts - Organic Chemistry
Card 1 of 1120
The molecule pictured is known as rapamycin, or Sirolimus, and is used as an immunosuppressant during organ transplants. Which of the following colored carbonyl groups is the most electrophilic?

The molecule pictured is known as rapamycin, or Sirolimus, and is used as an immunosuppressant during organ transplants. Which of the following colored carbonyl groups is the most electrophilic?

Tap to reveal answer
Note that many of these carbonyl groups are actually part of various functional groups. For example, the gold is an aldehyde, the green and purple are both ketones, the red is an amide, and the blue is an ester. We know the electrophilicity of carbonyl-containing functional groups is as follows:

Thus, our aldehyde, in gold, is the most electrophilic.
Note that many of these carbonyl groups are actually part of various functional groups. For example, the gold is an aldehyde, the green and purple are both ketones, the red is an amide, and the blue is an ester. We know the electrophilicity of carbonyl-containing functional groups is as follows:

Thus, our aldehyde, in gold, is the most electrophilic.
← Didn't Know|Knew It →
Which compound is not a Lewis acid?
Which compound is not a Lewis acid?
Tap to reveal answer
A Lewis acid is an electron pair acceptor. A Lewis base is an electron pair donor.
 is an ionic molecule and is neutral.
is an ionic molecule and is neutral.  and
and  have positive charges and are electron pair acceptors, as they are more stable when they are neutral.
have positive charges and are electron pair acceptors, as they are more stable when they are neutral.
The correct answer,  , has a lone pair on the nitrogen atom that can be donated to form bonds with other atoms, so it is a Lewis base.
, has a lone pair on the nitrogen atom that can be donated to form bonds with other atoms, so it is a Lewis base.
A Lewis acid is an electron pair acceptor. A Lewis base is an electron pair donor.



The correct answer, 
← Didn't Know|Knew It →
Which of these is a typical electrophile?
Which of these is a typical electrophile?
Tap to reveal answer
Electrophiles are substances that accept an electron pair to form a covalent bond, and nucleophiles are those that donate an electron pair to form a covalent bond. The chloride and iodide ions are both nucleophiles, as they each have a charge of  and would thus be willing to donate their extra electron. Ammonia (
and would thus be willing to donate their extra electron. Ammonia ( ) is also a nucleophile, as the nitrogen has a lone pair of electrons to donate. The methyl carbocation (carbon attached to three hydrogen atoms, with a positive charge) is an electrophile. The positive charge on the carbon makes it willing to accept an electron pair to form a covalent bond.
) is also a nucleophile, as the nitrogen has a lone pair of electrons to donate. The methyl carbocation (carbon attached to three hydrogen atoms, with a positive charge) is an electrophile. The positive charge on the carbon makes it willing to accept an electron pair to form a covalent bond.
Electrophiles are substances that accept an electron pair to form a covalent bond, and nucleophiles are those that donate an electron pair to form a covalent bond. The chloride and iodide ions are both nucleophiles, as they each have a charge of 

← Didn't Know|Knew It →
Which side (left or right) of the following reaction is favored and why?

Which side (left or right) of the following reaction is favored and why?

Tap to reveal answer
The side of the reaction that is favored will have the acid with the higher  , because the reaction goes (strong acid + strong base
, because the reaction goes (strong acid + strong base  weak acid + weak base).
weak acid + weak base).
The side of the reaction that is favored will have the acid with the higher 

← Didn't Know|Knew It →

For which of the following acid-base reactions will the equilibrium lie on the left side?
For which of the following acid-base reactions will the equilibrium lie on the left side?
Tap to reveal answer
The pKa value indicates how strong an acid is, and acid strength increases as pKa decreases. The side of a reaction with a lower pKa is going to dissociate more, pushing the equilibrium over to the other side. The equilibrium will thus lie on the side with the HIGHER pKa.

Since the pKa of acetic acid (4.76) is higher than the pKa of trifluoroacetic acid (0), the reaction will shift to the left to reach equilibrium.
The pKa value indicates how strong an acid is, and acid strength increases as pKa decreases. The side of a reaction with a lower pKa is going to dissociate more, pushing the equilibrium over to the other side. The equilibrium will thus lie on the side with the HIGHER pKa.
Since the pKa of acetic acid (4.76) is higher than the pKa of trifluoroacetic acid (0), the reaction will shift to the left to reach equilibrium.
← Didn't Know|Knew It →

Rank these weak acids by decreasing  (of the expressed hydrogens).
(of the expressed hydrogens).

Rank these weak acids by decreasing 
Tap to reveal answer
The governing principle regarding the prediction of  values (relative to other compounds) is to assess the stability of the product formed by the release of a proton. The release of the alkyne hydrogen in compound III results in a carbanion, a highly unstable species, so it is expected that this compound is the least acidic and has the highest
values (relative to other compounds) is to assess the stability of the product formed by the release of a proton. The release of the alkyne hydrogen in compound III results in a carbanion, a highly unstable species, so it is expected that this compound is the least acidic and has the highest  . Intuition serves well in this instance and we see that hydrogens bound to a triple bond have a
. Intuition serves well in this instance and we see that hydrogens bound to a triple bond have a  value of around 25. The relative stabilities of the remaining compounds may be assessed in the same manner. Compound IV is the second weakest acid
value of around 25. The relative stabilities of the remaining compounds may be assessed in the same manner. Compound IV is the second weakest acid  because the three methyl groups donate electron density such that if the oxygen is deprotonated, the resulting negative charge is destabilized. Methanol and water have a unique, non-intuitive relationship regarding their relative acidities. One would assume that water should be a stronger acid than other acids bound to alkyl groups (by the reasoning expressed for compound IV). This is the case for all alcohols except methanol, in which the delocalization of charge allowed by the increased molecular size outweighs the destabilization caused by electron donation. Thus methanol is a slightly stronger acid than water. This is evidenced in their
because the three methyl groups donate electron density such that if the oxygen is deprotonated, the resulting negative charge is destabilized. Methanol and water have a unique, non-intuitive relationship regarding their relative acidities. One would assume that water should be a stronger acid than other acids bound to alkyl groups (by the reasoning expressed for compound IV). This is the case for all alcohols except methanol, in which the delocalization of charge allowed by the increased molecular size outweighs the destabilization caused by electron donation. Thus methanol is a slightly stronger acid than water. This is evidenced in their  values: 15.7 for water and 15.5 for methanol. The correct ordering of the given compounds is: III, IV, II, I.
values: 15.7 for water and 15.5 for methanol. The correct ordering of the given compounds is: III, IV, II, I.
The governing principle regarding the prediction of 




← Didn't Know|Knew It →

List the given compounds in order of decreasing basicity.

List the given compounds in order of decreasing basicity.
Tap to reveal answer
An easy way to consider relative base strengths is to consider the strength of the compounds' conjugate acids.The stronger the conjugate acid, the weaker the base. Water (compound IV) is the least basic of the compounds because its conjugate acid,  is the strongest of the given compounds' conjugate acids
is the strongest of the given compounds' conjugate acids  . Carboxylate ions (compound III) are highly stabilized by resonance and predominate at neutral pH. The conjugate carboxylic acids readily donate protons (acetic acid:
. Carboxylate ions (compound III) are highly stabilized by resonance and predominate at neutral pH. The conjugate carboxylic acids readily donate protons (acetic acid:  ). Ammonia (compound I) has considerable basicity; binding a fourth hydrogen produces ammonium ion
). Ammonia (compound I) has considerable basicity; binding a fourth hydrogen produces ammonium ion  , which predominates at neutral pH
, which predominates at neutral pH  Sodium propoxide (compound II) is a strong base, bearing a full negative charge on its oxygen. Its conjugate acid, 1-propanol, is a rather weak acid
Sodium propoxide (compound II) is a strong base, bearing a full negative charge on its oxygen. Its conjugate acid, 1-propanol, is a rather weak acid  . Since its conjugate acid is the weakest (highest
. Since its conjugate acid is the weakest (highest  ), sodium propoxide is the strongest base. Based on the previous observations the correct ordering of the compounds is: II, I, III, IV.
), sodium propoxide is the strongest base. Based on the previous observations the correct ordering of the compounds is: II, I, III, IV.
An easy way to consider relative base strengths is to consider the strength of the compounds' conjugate acids.The stronger the conjugate acid, the weaker the base. Water (compound IV) is the least basic of the compounds because its conjugate acid, 






← Didn't Know|Knew It →
A carboxylic acid has a pKa of 5. At a pH of 8, what is the ratio of salt to acid?
A carboxylic acid has a pKa of 5. At a pH of 8, what is the ratio of salt to acid?
Tap to reveal answer
Use the Henderson Hasselbalch equation:
![pH = pKa + log $\frac{[salt]}{[acid]}$](https://vt-vtwa-assets.varsitytutors.com/vt-vtwa/uploads/formula_image/image/439185/gif.latex)
![8 = 5 + log $\frac{[salt]}{[acid]}$](https://vt-vtwa-assets.varsitytutors.com/vt-vtwa/uploads/formula_image/image/439186/gif.latex)


Use the Henderson Hasselbalch equation:
← Didn't Know|Knew It →
A.
B.
Which of the above molecules is expected to have a more acidic alpha-carbon, and why?
A.
B.
Which of the above molecules is expected to have a more acidic alpha-carbon, and why?
Tap to reveal answer
Molecule B will have a more acidic alpha-carbon because once the alpha proton becomes dissociated, the conjugate base will have relatively more stability than the conjugate base of molecule A.
When the alpha-carbon on molecule A loses it's proton, the conjugate base is not as stable. The reason for this is because the oxygen that is involved in the ester bond can contribute its electrons towards a resonance structure. Therefore, after the alpha-proton is lost, the alpha-carbon will have a negative charge that will be destabilized by the delocalized negative charge of the resonance structures.
Molecule B will have a more acidic alpha-carbon because once the alpha proton becomes dissociated, the conjugate base will have relatively more stability than the conjugate base of molecule A.
When the alpha-carbon on molecule A loses it's proton, the conjugate base is not as stable. The reason for this is because the oxygen that is involved in the ester bond can contribute its electrons towards a resonance structure. Therefore, after the alpha-proton is lost, the alpha-carbon will have a negative charge that will be destabilized by the delocalized negative charge of the resonance structures.
← Didn't Know|Knew It →
Which of the following is the strongest acid?
Which of the following is the strongest acid?
Tap to reveal answer
From the start, we know we can eliminate answer choice  because it is the only answer choice that is not a strong acid. Now we have three strong acids, but we have to determine which is strongest. To do so, we take the conjugate base of each strong acid to see which conjugate base is the weakest acid. Remember: weaker conjugate base means a stronger acid.
because it is the only answer choice that is not a strong acid. Now we have three strong acids, but we have to determine which is strongest. To do so, we take the conjugate base of each strong acid to see which conjugate base is the weakest acid. Remember: weaker conjugate base means a stronger acid.  is the largest ion of the bunch. Its large size allows it to better stabilize the negative charge and so it is the weakest (most stable) conjugate base. Because the weakest conjugate base leads to the strongest acid,
is the largest ion of the bunch. Its large size allows it to better stabilize the negative charge and so it is the weakest (most stable) conjugate base. Because the weakest conjugate base leads to the strongest acid,  is our correct answer.
is our correct answer.
From the start, we know we can eliminate answer choice 


← Didn't Know|Knew It →

Rank the given molecules in order of increasing pKa.

Rank the given molecules in order of increasing pKa.
Tap to reveal answer
Recall that the stronger an acid, the lower the pKa.
II (two fluorine atoms really close to  has largest inductive effect, so bond is most weakened, and pKa is lowest)
has largest inductive effect, so bond is most weakened, and pKa is lowest)
V (one fluorine really close to  has strong inductive effect)
has strong inductive effect)
I (one fluorine a little further away from  has weaker inductive effect)
has weaker inductive effect)
IV (no inductive effect)
III (alcohols are much less acidic than carboxylic acids, and it has the highest pKa of all)
Recall that the stronger an acid, the lower the pKa.
II (two fluorine atoms really close to 
V (one fluorine really close to 
I (one fluorine a little further away from 
IV (no inductive effect)
III (alcohols are much less acidic than carboxylic acids, and it has the highest pKa of all)
← Didn't Know|Knew It →
Which of the following sets of bases are listed from most basic to least basic?
Which of the following sets of bases are listed from most basic to least basic?
Tap to reveal answer
The correct ranking from most basic to least basic is:

The best bases are negatively charged, and the worst bases are positively charged (acidic). The stronger the base, the weaker (more stable) it's conjugate acid. An alkane is a very stable conjugate acid, which tells us that  is the most basic of the set.
is the most basic of the set.
We know based on charge alone, that  is more basic than
is more basic than  but less basic than
but less basic than  or
or  .
.
We know that  is more basic
is more basic  because the electronegativity of
because the electronegativity of  is less than that of
is less than that of  . This means the lone pair of electrons on
. This means the lone pair of electrons on  are held less tightly and more likely to pick up a proton.
are held less tightly and more likely to pick up a proton.
The correct ranking from most basic to least basic is:
The best bases are negatively charged, and the worst bases are positively charged (acidic). The stronger the base, the weaker (more stable) it's conjugate acid. An alkane is a very stable conjugate acid, which tells us that 
We know based on charge alone, that 



We know that 




← Didn't Know|Knew It →
Which of the following sets of acids are correctly listed from most to least acidic?
Which of the following sets of acids are correctly listed from most to least acidic?
Tap to reveal answer
Remember that the strongest acids have the weakest conjugate bases.  is more acidic than
is more acidic than  because iodine has a larger atomic radius than bromine.
because iodine has a larger atomic radius than bromine.  ,
,  , and
, and  are strong acids and should be at the beginning of the list. Alkanes are not acidic. Acetic acid is a weak acid (pKa =4-5).
are strong acids and should be at the beginning of the list. Alkanes are not acidic. Acetic acid is a weak acid (pKa =4-5).
Remember that the strongest acids have the weakest conjugate bases. 




← Didn't Know|Knew It →
Chemists and biochemists have many ways of representing sugars. Glucose, the most common hexose, is shown below in various linear and cyclic projections. Using the linear and cyclic projection of your choice, can you indicate which colored oxygen in the linear form corresponds to the circled hemiacetal oxygen once the cyclization reaction is complete?
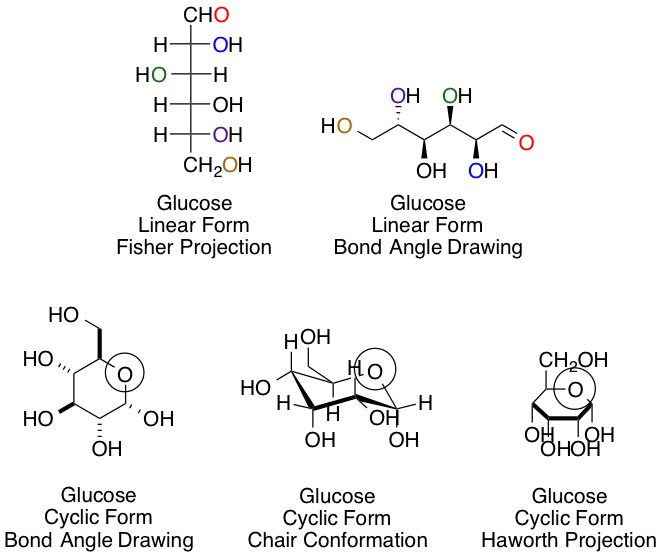
Chemists and biochemists have many ways of representing sugars. Glucose, the most common hexose, is shown below in various linear and cyclic projections. Using the linear and cyclic projection of your choice, can you indicate which colored oxygen in the linear form corresponds to the circled hemiacetal oxygen once the cyclization reaction is complete?

Tap to reveal answer
This answer, regardless of your preference of projection type, is easiest to obtain using arrow pushing for the cyclization reaction to keep track of each carbon and oxygen:

The purple carbon in the linear projection ends in the circled hemiacetal position.
This answer, regardless of your preference of projection type, is easiest to obtain using arrow pushing for the cyclization reaction to keep track of each carbon and oxygen:

The purple carbon in the linear projection ends in the circled hemiacetal position.
← Didn't Know|Knew It →
Which of the following structures represents the anomeric alpha ring structure of D-glucose?

Which of the following structures represents the anomeric alpha ring structure of D-glucose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-glucose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and trans with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and trans with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-glucose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Which of the following structures represents the anomeric alpha ring structure of D-galactose?

Which of the following structures represents the anomeric alpha ring structure of D-galactose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-galactose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and cis with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be trans, cis, and cis with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-galactose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Which of the following ring structures represents the anomeric alpha ring structure of D-mannose?

Which of the following ring structures represents the anomeric alpha ring structure of D-mannose?

Tap to reveal answer
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the  attached to carbon 5, while those that are in the left position end up cis to the
attached to carbon 5, while those that are in the left position end up cis to the  attached to carbon 5.
attached to carbon 5.
If the hydroxyl group attached to carbon 1 ends up trans to the  attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the
attached to carbon 5, the ring structure is considered alpha. If the hydroxyl group attached to carbon 1 is cis to the  attached to carbon 5, the ring structure is considered beta.
attached to carbon 5, the ring structure is considered beta.
The alpha ring structure of D-mannose bonds the carbon 1 hydroxyl group trans to the carbon 5  group. The hyroxyl groups on carbons 2, 3, and 4 will be cis, cis, and trans with respect to the
group. The hyroxyl groups on carbons 2, 3, and 4 will be cis, cis, and trans with respect to the  .
.
When converting a linear sugar to its ring form, a bond is formed between the oxygen attached to carbon 5 and the carbon at position 1. All hydroxyl groups that are not attached to the carbon in position 1 and are oriented to the right end up trans to the 

If the hydroxyl group attached to carbon 1 ends up trans to the 

The alpha ring structure of D-mannose bonds the carbon 1 hydroxyl group trans to the carbon 5 

← Didn't Know|Knew It →
Identify the aldose pictured, including its alpha or beta designation.

Identify the aldose pictured, including its alpha or beta designation.

Tap to reveal answer
The structure pictured is mannose because the hydroxyl groups at carbons 2, 3, and 4 are situated cis, cis, and trans (respectively) to the  attached to carbon 5.
attached to carbon 5.
The mannose pictured is in alpha form because the hydroxyl group at carbon 1 is trans to the  attached to carbon 5.
attached to carbon 5.
The structure pictured is mannose because the hydroxyl groups at carbons 2, 3, and 4 are situated cis, cis, and trans (respectively) to the 
The mannose pictured is in alpha form because the hydroxyl group at carbon 1 is trans to the 
← Didn't Know|Knew It →
The Fischer projection pictured is a form of glucose. The carbon labeled "x" is the chiral carbon farthest away from carbon 1 and the hydroxyl group connected to carbon "x" is on the right. This fact designates that the glucose as what configuration?

The Fischer projection pictured is a form of glucose. The carbon labeled "x" is the chiral carbon farthest away from carbon 1 and the hydroxyl group connected to carbon "x" is on the right. This fact designates that the glucose as what configuration?

Tap to reveal answer
The chiral carbon farthest away from carbon 1 is designated as "D" if its hydroxyl group is on the right side in the Fischer projection. In other words, this is D-glucose because the hyroxyl group on carbon "x" is oriented to the right.
The chiral carbon farthest away from carbon 1 is designated as "D" if its hydroxyl group is on the right side in the Fischer projection. In other words, this is D-glucose because the hyroxyl group on carbon "x" is oriented to the right.
← Didn't Know|Knew It →
What is the name of the aldose pictured in this Fischer projection?

What is the name of the aldose pictured in this Fischer projection?

Tap to reveal answer
The structure is D-ribose because it is a five-carbon aldose with the hydroxyl groups on carbons 2, 3, and 4 all on the right in the Fischer projection.
The structure is D-ribose because it is a five-carbon aldose with the hydroxyl groups on carbons 2, 3, and 4 all on the right in the Fischer projection.
← Didn't Know|Knew It →