All Organic Chemistry Resources
Example Questions
Example Question #11 : Organic Oxidizing Agents
A chemist adds the orange oxidizing agent, Na2Cr2O7, to the following substrates and dissolves the mixture in an aqueous solution of sulfuric acid. Oxidation is indicated by the disappearance of the orange color. Which of the substrate-oxidant solutions will remain orange?
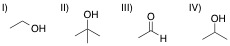
II
IV
All of the solutions will lose their orange color, indicating oxidation
III
I
II
The following reaction schemes show the oxidation of all substrates, indicating that substrate II is in the highest oxidation state possible, and that an oxidation of this compound will not proceed.
Remember that in sulfuric acid and water, Na2Cr2O7 will be converted to CrO3, the active oxidant species. Furthermore, the oxidation mechanism involving this species includes the key step in which a hydrogen bonded to the carbon in question is eliminated, and simutaneously, a double bond from that carbon to an oxygen is installed. Thus, all substrates that feature at least one hydrogen bonded to the carbon to be oxidized can and will be oxidized in the precense of chromium trioxide.
Lastly, remember that these reactions are taking place in the prescence of water. While substrates such as compound III do not appear to be oxidizable, attack of water at the aldehyde carbon will give a dialcohol tetrathedral intermediate that can be immediately oxidized by chromium trioxide to the corresponding carboxylic acid. A similar mechanism occurs for substrate I, wherein, after the ketone oxidation state is achieved, an attack of water furnishes the same dialcohol intermediate that is oxidized to the carboxylic acid. Remember that the highest oxidation state available for organic compounds containing more than one carbon is the carboxylic acid oxidation state. Chromium trioxide will oxidize all organics to this oxidation state, unless directly-bonded hydrogens are not present in lower oxidation states, such as shown with substrate IV.

Example Question #12 : Organic Oxidizing Agents
Which of the following reagents would satisfy the given reaction?
In order to drive the reactant, we must first convert the alcohol group on the ethanol into a carboxylic acid. We do so by using the oxidizing agent, 

Example Question #13 : Redox Chemistry
Which of the following substrates will be oxidized into a ketone when reacting with 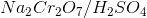
A primary alcohol
An aldehyde
A carboxylic acid
A secondary alcohol
A secondary alcohol

Not only can 
Example Question #2 : Using Dichromate Compounds

What would be the product of the given reaction?


![]()
![]()

The reaction given would give a ketone. This type of reaction is called an oxidation reaction. Oxidation of a secondary alcohol as in the reaction given by 

Certified Tutor
All Organic Chemistry Resources













