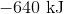All SAT II Chemistry Resources
Example Questions
Example Question #1 : Enthalpy
Suppose that 1.5 moles of potassium reacts completely with an excess of chlorine gas. What is the enthalpy change of the reaction?
Possible Answers:
Correct answer:
Explanation:
First, note that heat is a product of the reaction. This means that the reaction releases heat and is exothermic, so the enthalpy change must be negative. Next, consider that 2 moles of sodium would react to produce 640 kJ. Thus, 1.5 moles of sodium would react to produce

Hence, the enthalpy change is 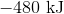
Bhavi
Certified Tutor
Certified Tutor
Touro College, Master of Science, Biological and Physical Sciences. Touro College, Doctor of Medicine, Osteopathic Medicine (...
Brianna
Certified Tutor
Certified Tutor
Fairfield University, Bachelor of Science, Biology, General. National University of Ireland at Galway, Master of Science, Neu...
All SAT II Chemistry Resources
Popular Subjects
Statistics Tutors in Dallas Fort Worth, Math Tutors in Boston, GRE Tutors in Atlanta, Reading Tutors in New York City, Spanish Tutors in Boston, Biology Tutors in Philadelphia, Statistics Tutors in San Diego, Calculus Tutors in Philadelphia, Physics Tutors in San Diego, Algebra Tutors in San Francisco-Bay Area
Popular Courses & Classes
SAT Courses & Classes in Phoenix, SSAT Courses & Classes in Philadelphia, ISEE Courses & Classes in Atlanta, GMAT Courses & Classes in Washington DC, ACT Courses & Classes in Atlanta, GRE Courses & Classes in Atlanta, GRE Courses & Classes in Phoenix, SAT Courses & Classes in Houston, GRE Courses & Classes in Philadelphia, Spanish Courses & Classes in Houston