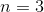All GRE Subject Test: Chemistry Resources
Example Questions
Example Question #2 : Help With Alkene Synthesis

What is the value of 
Possible Answers:
Correct answer:
Explanation:
Huckel's rule states that an aromatic compound must have 
If 4n+2=18, then n=4.
Leann
Certified Tutor
Certified Tutor
Georgia Institute of Technology-Main Campus, Engineer, Systems Engineering. University of South Florida-Main Campus, Masters ...
All GRE Subject Test: Chemistry Resources
Popular Subjects
ACT Tutors in Denver, Computer Science Tutors in Boston, ACT Tutors in Phoenix, MCAT Tutors in Chicago, MCAT Tutors in Boston, LSAT Tutors in Boston, Calculus Tutors in New York City, Computer Science Tutors in Houston, Calculus Tutors in Dallas Fort Worth, Chemistry Tutors in Chicago
Popular Courses & Classes
GRE Courses & Classes in New York City, ACT Courses & Classes in Los Angeles, MCAT Courses & Classes in Washington DC, ISEE Courses & Classes in Houston, ACT Courses & Classes in Houston, GRE Courses & Classes in Boston, Spanish Courses & Classes in Los Angeles, ACT Courses & Classes in San Diego, LSAT Courses & Classes in San Francisco-Bay Area, GMAT Courses & Classes in Atlanta
Popular Test Prep
MCAT Test Prep in New York City, ISEE Test Prep in Washington DC, ACT Test Prep in Dallas Fort Worth, LSAT Test Prep in San Francisco-Bay Area, ACT Test Prep in San Diego, GMAT Test Prep in Los Angeles, MCAT Test Prep in San Francisco-Bay Area, LSAT Test Prep in Denver, SSAT Test Prep in Houston, GRE Test Prep in Los Angeles