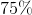All High School Physics Resources
Example Questions
Example Question #1 : Heat
A Carnot Cycle operates using a heat reservoir at a temperature of 


Possible Answers:
Correct answer:
Explanation:
The efficiency of a Carnot Cycle is defined by:
In this calculation, temperatures are given in Kelvin. Since Celsius and Kelvin exist in a one-to-one ratio, a conversion is unnecessary unless desired. We will perform the conversion, since most classes will advise it.
Using these values, we can calculate the efficiency:
Convert this value to a percentage:
All High School Physics Resources
Popular Subjects
Math Tutors in Phoenix, Biology Tutors in Dallas Fort Worth, Reading Tutors in Philadelphia, Math Tutors in Denver, Reading Tutors in Denver, Chemistry Tutors in Miami, Calculus Tutors in San Diego, ISEE Tutors in San Francisco-Bay Area, SSAT Tutors in San Diego, Algebra Tutors in Philadelphia
Popular Courses & Classes
Spanish Courses & Classes in San Diego, LSAT Courses & Classes in Phoenix, SSAT Courses & Classes in Miami, MCAT Courses & Classes in Houston, GMAT Courses & Classes in Houston, SAT Courses & Classes in Dallas Fort Worth, GMAT Courses & Classes in Los Angeles, GRE Courses & Classes in Los Angeles, GRE Courses & Classes in San Francisco-Bay Area, GMAT Courses & Classes in Denver
Popular Test Prep
MCAT Test Prep in Denver, LSAT Test Prep in Phoenix, GMAT Test Prep in Los Angeles, GRE Test Prep in Philadelphia, GMAT Test Prep in Atlanta, SSAT Test Prep in San Diego, MCAT Test Prep in San Francisco-Bay Area, SAT Test Prep in Denver, GRE Test Prep in Chicago, MCAT Test Prep in New York City